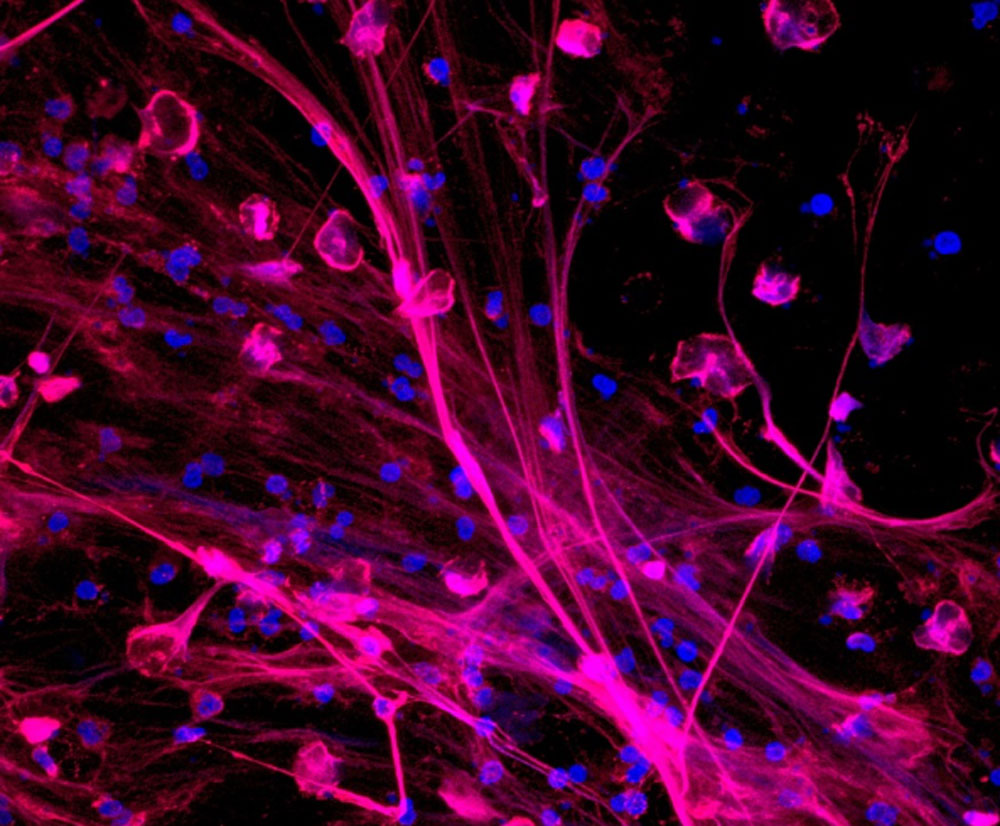
 Neutrophils releasing neutrophil extracellular traps (NETs). NETs are stained to visualize neutrophil myeloperoxidase (red) and DNA (blue). Image courtesy Zdenek Hel, Ph.D.It is the stuff of nightmares. Patients suffering from acute respiratory distress syndrome (ARDS) during COVID-19 say it felt like “I was gasping my last breath“ and “as if somebody had taken a scuba diving tank while I'm underwater and turned it off completely.”
Neutrophils releasing neutrophil extracellular traps (NETs). NETs are stained to visualize neutrophil myeloperoxidase (red) and DNA (blue). Image courtesy Zdenek Hel, Ph.D.It is the stuff of nightmares. Patients suffering from acute respiratory distress syndrome (ARDS) during COVID-19 say it felt like “I was gasping my last breath“ and “as if somebody had taken a scuba diving tank while I'm underwater and turned it off completely.”
The development of ARDS is a predictor of high mortality in COVID-19 patients. But clinicians do not currently have a reliable method to predict which patients will develop ARDS.
A group of UAB researchers have a hypothesis, however. Tracking the emergence of specific subtypes of immune cells as they respond to SARS-CoV-2, the virus that causes COVID-19, could help predict the emergence of ARDS, the scientists suspect. Their work is funded through a pilot grant from UAB's urgent COVID-19 research fund.
Zdenek Hel, Ph.D., professor in the Division of Molecular and Cellular Pathology, is the principal investigator for the new project, “Neutrophils as a driving mechanism of acute respiratory distress syndrome and death in COVID-19 patients.” His collaborators are Paul Goepfert, M.D., and Turner Overton, M.D., both professors in the Division of Infectious Diseases.
 Zdenek Hel, Ph.D.Neutrophils are the first responders of the immune system. They are everywhere: More than 100 billion neutrophils are produced every day. (With a half-life of 18 to 19 hours, they must be continuously replenished.)
Zdenek Hel, Ph.D.Neutrophils are the first responders of the immune system. They are everywhere: More than 100 billion neutrophils are produced every day. (With a half-life of 18 to 19 hours, they must be continuously replenished.)
Traditionally, neutrophils were thought to wipe out invading pathogens by eating them (phagocytosis) or using highly toxic, bleach-like granules to destroy the cell walls of foreign microorganisms. But in 2004, immunologists discovered a third strategy: neutrophil extracellular traps, or NETs. These are a meshwork of DNA-based fibers that some neutrophils expel to immobilize and kill invading microorganisms such as viruses and bacteria.
But the generation of this entrapment mechanism, known as NETosis, recruits other neutrophils and additional immune cells to the area in a pro-inflammatory process. This accumulation of immune cells seems to amplify NET production and is thought to be a major cause of lung-tissue injury and breathing problems seen in ARDS.
“We hypothesize that in certain conditions, such as COVID-19, populations of neutrophils arise that have a higher capacity to product NETs,” Hel said.
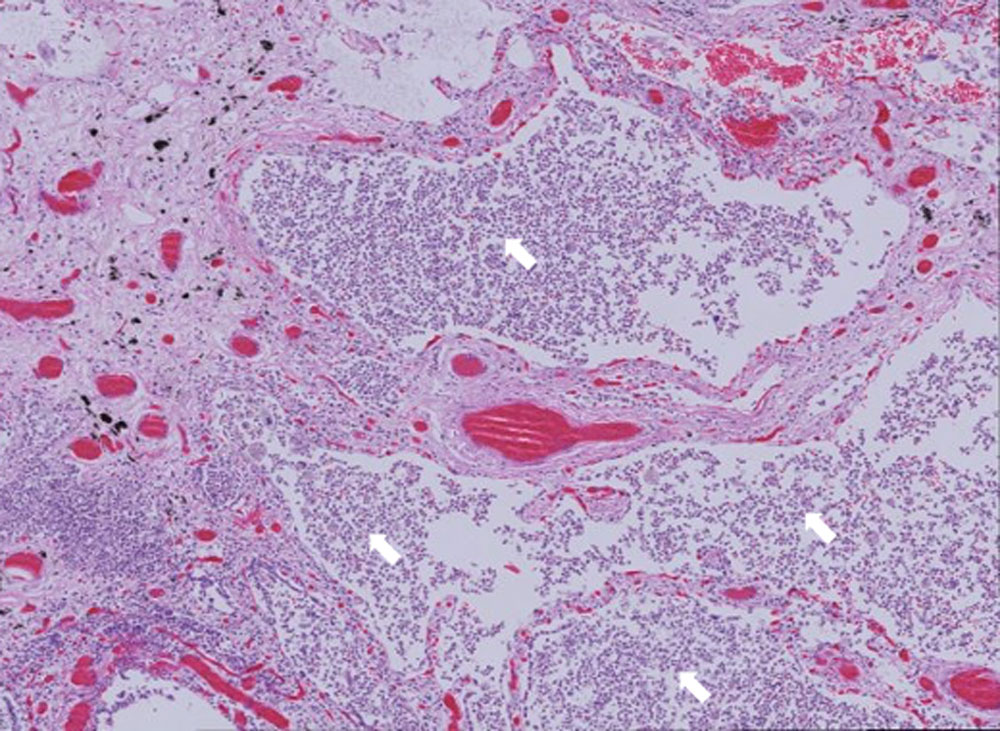 Neutrophil accumulation in the lung alveoli in an autopsy sample from a patient who succumbed to COVID-19.Image courtesy Zdenek Hel, Ph.D.
Neutrophil accumulation in the lung alveoli in an autopsy sample from a patient who succumbed to COVID-19.Image courtesy Zdenek Hel, Ph.D.
The immune mechanisms underlying ARDS are not well understood, the researchers noted in their project summary, although multiple studies have demonstrated an essential role for neutrophils in the syndrome and severe cases of COVID-19. “In collaboration with Dr. Benson and other colleagues in the Department of Pathology, we have identified a large accumulation of neutrophils in the lungs of COVID-19 patients,” Hel said, “and our preliminary data show a significant perturbation of the phenotype of circulating neutrophils suggesting pathological properties.”
“I am convinced that, once we learn how to regulate neutrophil activity, we will gain access to powerful new treatments of COVID-19 and other respiratory diseases.” |
Their project has three aims. First, the researchers will determine whether the emergence of pathological neutrophil populations and elevation of plasma markers of NETosis — such as DNA associated with myeloperoxidase — are predictive of disease severity. Then they will characterize the properties of neutrophil populations in SARS-CoV-2 infections. Finally, they will use a form of single-cell sequencing, known as CITE-Seq, “to determine the frequency, activation status and specific properties of immune populations in the blood of SARS-CoV-2-infected individuals,” they write.
“CITE-Seq is an amazing and powerful new technology,” Hel said. “To my knowledge, our lab was the first at UAB to use it, although there may be more groups now.” It uses the 10X Genomics platform available in UAB's Single Cell Sequencing Core Facility. “However, it adds antibodies connected to short strands of DNA,” Hel said. “In this way, it provides the information on RNA expression inside the individual cell as well as the type and level of protein present on the cell surface.”
“A clear definition of the role of neutrophils in SARS-CoV-2 infections will result in the design of new therapeutic approaches for pharmacological targeting neutrophil dysregulation and development,” Hel and his colleagues write.
“Neutrophil activation and NETosis can be pharmacologically modulated by targeting NADPH oxidase, colony-stimulating factors and receptors, or CXCR2 chemokine receptor,” Hel said. “Various promising modulators of neutrophil recruitment and activation are currently in Phase II and III clinical studies. I am convinced that, once we learn how to regulate neutrophil activity, we will gain access to powerful new treatments of COVID-19 and other respiratory diseases.”