 The University of Alabama at Birmingham Comprehensive Cancer Center is now enrolling patients for the National Cancer Institute Molecular Analysis for Therapy Choice, or NCI-MATCH study, the largest, most scientifically rigorous precision medicine trial in cancer to date.
The University of Alabama at Birmingham Comprehensive Cancer Center is now enrolling patients for the National Cancer Institute Molecular Analysis for Therapy Choice, or NCI-MATCH study, the largest, most scientifically rigorous precision medicine trial in cancer to date.
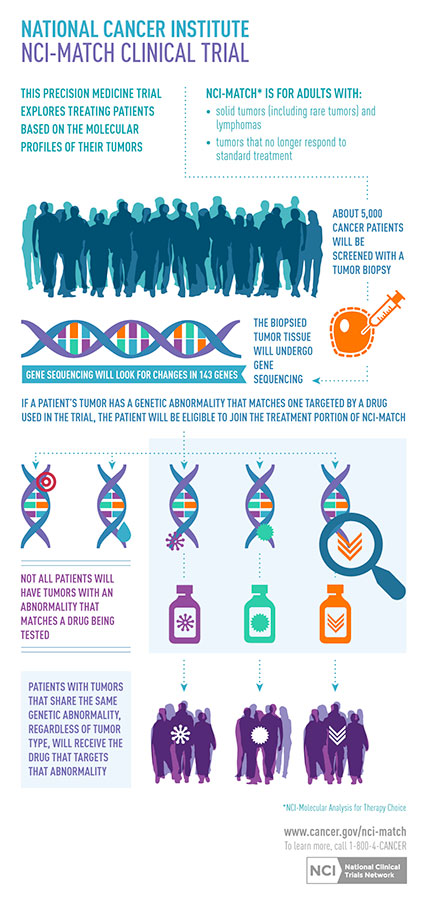
Precision medicine is a medical model that customizes treatment to the individual patient; this study will provide targeted therapy for cancer patients, while measuring effectiveness in a variety of cancers. This clinical trial is available for patients who have cancer that has returned or gotten worse after standard treatment, or a type of cancer that has no standard treatment.
Patients with the same type and stage of cancer usually receive the same cancer treatments. However, patients can respond differently to these treatments. In learning more about cancer, researchers have discovered that genetic changes, or molecular abnormalities, in a patient’s tumor may cause the cancer to grow.
“This research study, commonly known as a clinical trial, is an example of what precision medicine is all about,” said Carla Falkson, M.D., professor in the UAB Division of Hematology and Oncology and principal investigator for the trial. “We are at an exciting point in cancer where we can tailor care to the individual patient based on emerging treatment and prevention that takes into account individual variability in genes, environment and lifestyle for each person.”
The NCI-MATCH will assign, or “match,” a patient to a specific cancer treatment based on whether certain genetic changes are found in their tumor. The tests that are done on the tumor to find out whether a patient has genetic changes or mutations are investigational. If the patient’s tumor has genetic changes that match one of the study drugs being studied in the trial, the patient can take part.
Patients will have to undergo a biopsy and may have to have other tests to see whether they can be in the treatment part of the study. Even if patients have had a biopsy before, they will need another biopsy to determine new genetic changes.
“Overall, this study will help researchers learn whether study drugs targeting certain genetic changes in cancer cells can be effective in treating a variety of cancers,” said Falkson, a senior scientist in the experimental therapeutics program at the UAB Comprehensive Cancer Center. “This type of clinical trial is very broad in nature and has the capability of enrolling many patients locally, regionally and nationally; the information we learn from them can save countless lives.”
Overall, the trial seeks to enroll 3,000 adults who are 18 years of age or older. Initially, the trial will be open with 10 treatments, and then an additional 12 treatments are expected to be added. There will be 35 patients enrolled for each drug/drug combination being studied.
Study drugs have been approved by the United States Food and Drug Administration for a specific type of cancer with a particular genetic change. Other drugs are still being studied, but have shown some effect across different cancers with a specific genetic change.
“When patients participate in a clinical trial, they are really participating in their own care,” said Edward Partridge, M.D., director of the UAB Comprehensive Cancer Center. “A clinical trial is really the bridge between research and patient care. We encourage participation in them because we want our patients to get the most innovative and advanced care possible for their specific cancer.”
The UAB Comprehensive Cancer Center was recently selected as one of only 30 institutions in the United States to receive funding as a Lead Academic Participating Site for the NCI’s newly created National Clinical Trials Network. “This is an outstanding achievement, and we are incredibly excited to have this opportunity to expand our clinical trials enterprise on a more national level,” Partridge said.
The study was co-developed by ECOG-ACRIN Cancer Research Group and the NCI. ECOG-ACRIN coordinates the genetic testing. It also supports all trial sites with training, laboratory services, trial assignments, biostatistical support, data management, auditing, quality control and public awareness.
Patients with a cancer diagnosis are encouraged to talk to their doctors about the NCI-MATCH study and whether it may be appropriate for them.
For detailed information and eligibility requirements for the NCI-MATCH study, contact Liz Busby, director of Oncology Clinical Trials at the UAB Comprehensive Cancer Center, at 205-934-0337 or lizbusby@uab.edu.